
JosD2, Active
Recombinant full-length human JosD2 was expressed in E. coli cells using an N-terminal His tag.
Catalog No. I560-380H
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| I560-380H-05 | 5 ug | $226 | |
| I560-380H-10 | 10 ug | $325 | |
| I560-380H-BULK | BULK | Contact Us |

Recombinant full-length human JosD2 was expressed in E. coli cells using an N-terminal His tag.
Catalog No. I560-380H
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| I560-380H-05 | 5 ug | $226 | |
| I560-380H-10 | 10 ug | $325 | |
| I560-380H-BULK | BULK | Contact Us |
Overview:
JosD2 (Josephin domain-containing 2) is a member of the MJD family of deubiquitinating enzymes (DUBs) that is localized to the cytoplasm and catalyzes the hydrolysis of the bond between the C-terminal glycine of the ubiquitin peptide and protein substrates. In vitro, JosD2 can cleave ubiquitin chains (1). Although JosD2 has been shown to have deubiquitinating activities, details of its substrate specificity are still mainly unknown (2).
Gene Aliases:
SBBI54
Genbank Number:
References:
1. Seki, T. et al: JosD1, a membrane-targeted deubiquitinating enzyme, is activated by ubiquitination and regulates membrane dynamics, cell motility, and endocytosis. Journal of biological chemistry 288(24): 17145-55, 2013.
2. Tzvetkov, N. & Breuer, P: Josephin domain-containing proteins from a variety of species are active de-ubiquitination enzymes. Biol Chem 388(9):973-8, 2007.
Specific Activity:
Sample Activity Plot. For specific information on a given lot, see related technical data sheet.
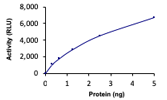
|
Purity:
Sample Purity Data. For specific information on a given lot, see related technical data sheet.

|
Storage, Stability, and Shipping:
Store product at –70oC. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Molecular Weight:
20 kDa
There are no related publications available for this product.
Cancer, Cardiovascular Disease, Cell Cycle, Cellular Stress, Invasion/Metastasis, Metabolic Disorder
STAY CONNECTED
Fax: 1-604-232-4601