
DAPK3, Active
Full-length recombinant human DAPK3 was expressed by baculovirus in Sf9 insect cells using an N-terminal GST tag.
Catalog No. D03-10G
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| D03-10G-05 | 5 ug | $226 | |
| D03-10G-10 | 10 ug | $325 | |
| D03-10G-BULK | BULK | Contact Us |
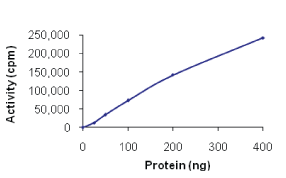

 DAPK1, Active, D01-11G
DAPK1, Active, D01-11G


 Toll Free: 1-866-954-6273
Toll Free: 1-866-954-6273 info@signalchem.com
info@signalchem.com