
Tau-441 Protein
Recombinant full-length human tag-free Tau-441 was expressed in E. coli cells.
Catalog No. T08-54N
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| T08-54N-20 | 20 ug | $160 | |
| T08-54N-50 | 50 ug | $336 | |
| T08-54N-BULK | BULK | Contact Us |
Home > Proteins > Tau Proteins

Recombinant full-length human tag-free Tau-441 was expressed in E. coli cells.
Catalog No. T08-54N
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| T08-54N-20 | 20 ug | $160 | |
| T08-54N-50 | 50 ug | $336 | |
| T08-54N-BULK | BULK | Contact Us |
Overview:
Tau-441 or Tau-F is a member of the Tau family of proteins which function to stabilize the microtubules by binding to them. Tau proteins are subject to phosphorylation and this phenomenon regulates the association of the Tau protein with the microtubules (1). Deposits of Alzheimer's disease AD-associated proteins, such as hyperphosphorylated Tau, as well as other shared misfolded proteins, such as, β-amyloid precursor protein (βAPP), ubiquitin, and various chaperones and protein kinases are thought to play a pathologic role in the cognitive decline and muscular failure. Malfunctioning of Tau proteins is associated with microtubules disintegration and collapsing of the neuronal transport system (2).
Gene Aliases:
Tau-F, (2N4R), Tau-4, MAPT, MSTD; PPND; DDPAC; MAPTL; MTBT1; MTBT2; FTDP-17; FLJ31424; MGC138549
Genbank Number:
References:
1. Zilka, N., et al. Truncated tau from sporadic Alzheimer's disease suffices to drive neurofibrillary degeneration in vivo. FEBS Lett. 2006; 508: 3582-3588.
2. Rial, A. et al: Calcium Dyshomeostasis in β-Amyloid and Tau-bearing Skeletal Myotubes. J. Biol. Chem., 2004; 279: 3524-53532.
Purity:
Sample Purity Data. For specific information on a given lot, see related technical data sheet.
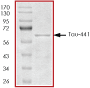
|
Storage, Stability and Shipping:
Store product at –70oC. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Molecular Weight:
~64 kDa
![]() Peng Z et al., A droplet reactor on a super-hydrophobic surface allows control and characterization of amyloid fibril growth Communications Biology September 2020 10.1038/s42003-020-01187-7
Peng Z et al., A droplet reactor on a super-hydrophobic surface allows control and characterization of amyloid fibril growth Communications Biology September 2020 10.1038/s42003-020-01187-7
![]() SH Lee et al., Antibody-Mediated Targeting of Tau In Vivo Does Not Require Effector Function and Microglial Engagement. Cell Reports August 2016 10.1016/j.celrep.2016.06.099
SH Lee et al., Antibody-Mediated Targeting of Tau In Vivo Does Not Require Effector Function and Microglial Engagement. Cell Reports August 2016 10.1016/j.celrep.2016.06.099
![]() A Chakraborty et al., Inositol hexakisphosphate kinase-1 regulates behavioral responses via GSK3 signaling pathways. Molecular Psychiatry March 2014 10.1038/mp.2013.21
A Chakraborty et al., Inositol hexakisphosphate kinase-1 regulates behavioral responses via GSK3 signaling pathways. Molecular Psychiatry March 2014 10.1038/mp.2013.21
![]() Absalon Sabrina et al., MiR-26b, Upregulated in Alzheimer's Disease, Activates Cell Cycle Entry, Tau-Phosphorylation, and Apoptosis in Postmitotic Neurons Journal of Neuroscience September 2013 10.1523/JNEUROSCI.1327-13.2013
Absalon Sabrina et al., MiR-26b, Upregulated in Alzheimer's Disease, Activates Cell Cycle Entry, Tau-Phosphorylation, and Apoptosis in Postmitotic Neurons Journal of Neuroscience September 2013 10.1523/JNEUROSCI.1327-13.2013
![]() Kawakami Fumitaka et al., Leucine-rich repeat kinase 2 regulates tau phosphorylation through direct activation of glycogen synthase kinase-3β FEBS Journal November 2013 10.1111/febs.12579
Kawakami Fumitaka et al., Leucine-rich repeat kinase 2 regulates tau phosphorylation through direct activation of glycogen synthase kinase-3β FEBS Journal November 2013 10.1111/febs.12579
![]() Kawakami Fumitaka et al., Stimulatory effect of α-synuclein on the tau-phosphorylation by GSK-3β FEBS Journal October 2011 10.1111/j.1742-4658.2011.08389.x
Kawakami Fumitaka et al., Stimulatory effect of α-synuclein on the tau-phosphorylation by GSK-3β FEBS Journal October 2011 10.1111/j.1742-4658.2011.08389.x
![]() Coutadeur Severine et al., A novel DYRK1A (Dual specificity tyrosine phosphorylation-regulated kinase 1A) inhibitor for the treatment of Alzheimer's disease: effect on Tau and amyloid pathologies in vitro Journal of Neurochemistry May 2015 10.1111/jnc.13018
Coutadeur Severine et al., A novel DYRK1A (Dual specificity tyrosine phosphorylation-regulated kinase 1A) inhibitor for the treatment of Alzheimer's disease: effect on Tau and amyloid pathologies in vitro Journal of Neurochemistry May 2015 10.1111/jnc.13018
![]() CJ Dunning et al., Direct High Affinity Interaction between Aβ42 and GSK3α Stimulates Hyperphosphorylation of Tau. A New Molecular Link in Alzheimer's Disease? ACS Chemical Neuroscience February 2016 10.1021/acschemneuro.5b00262
CJ Dunning et al., Direct High Affinity Interaction between Aβ42 and GSK3α Stimulates Hyperphosphorylation of Tau. A New Molecular Link in Alzheimer's Disease? ACS Chemical Neuroscience February 2016 10.1021/acschemneuro.5b00262
![]() Kawakami Fumitaka et al., LRRK2 Phosphorylates Tubulin-Associated Tau but Not the Free Molecule: LRRK2-Mediated Regulation of the Tau-Tubulin Association and Neurite Outgrowth PLoS One January 2012 10.1371/journal.pone.0030834
Kawakami Fumitaka et al., LRRK2 Phosphorylates Tubulin-Associated Tau but Not the Free Molecule: LRRK2-Mediated Regulation of the Tau-Tubulin Association and Neurite Outgrowth PLoS One January 2012 10.1371/journal.pone.0030834
![]() Frost Danielle et al., β-Carboline Compounds, Including Harmine, Inhibit DYRK1A and Tau Phosphorylation at Multiple Alzheimer's Disease-Related Sites PLoS One May 2011 10.1371/journal.pone.0019264
Frost Danielle et al., β-Carboline Compounds, Including Harmine, Inhibit DYRK1A and Tau Phosphorylation at Multiple Alzheimer's Disease-Related Sites PLoS One May 2011 10.1371/journal.pone.0019264
![]() et al., N-terminal and mid-region tau fragments as fluid biomarkers in neurological diseases Brain August 2022
et al., N-terminal and mid-region tau fragments as fluid biomarkers in neurological diseases Brain August 2022
![]() et al., Cerebrospinal fluid p-tau231 as an early indicator of emerging pathology in Alzheimer's disease EBioMedicine February 2022
et al., Cerebrospinal fluid p-tau231 as an early indicator of emerging pathology in Alzheimer's disease EBioMedicine February 2022
![]() et al., Tau activates microglia via the PQBP1-cGAS-STING pathway to promote brain inflammation Nature Communications November 2021
et al., Tau activates microglia via the PQBP1-cGAS-STING pathway to promote brain inflammation Nature Communications November 2021
![]() et al., P?tau235: a novel biomarker for staging preclinical Alzheimer�s disease EMBO Molecular Medicine November 2021
et al., P?tau235: a novel biomarker for staging preclinical Alzheimer�s disease EMBO Molecular Medicine November 2021
![]() et al., Prediction of future Alzheimer's disease dementia using plasma phospho-tau combined with other accessible measures. Nature medicine August 2021
et al., Prediction of future Alzheimer's disease dementia using plasma phospho-tau combined with other accessible measures. Nature medicine August 2021
![]() et al., Population-based blood screening for preclinical Alzheimer�s disease in a British birth cohort at age 70 Brain February 2021
et al., Population-based blood screening for preclinical Alzheimer�s disease in a British birth cohort at age 70 Brain February 2021
![]() et al., Head?to?head comparison of clinical performance of CSF phospho?tau T181 and T217 biomarkers for Alzheimer's disease diagnosis Alzheimer's & Dementia November 2020
et al., Head?to?head comparison of clinical performance of CSF phospho?tau T181 and T217 biomarkers for Alzheimer's disease diagnosis Alzheimer's & Dementia November 2020
![]() et al., Novel tau biomarkers phosphorylated at T181, T217 or T231 rise in the initial stages of the preclinical Alzheimer�s continuum when only subtle changes in A? pathology are detected EMBO Molecular Medicine November 2020
et al., Novel tau biomarkers phosphorylated at T181, T217 or T231 rise in the initial stages of the preclinical Alzheimer�s continuum when only subtle changes in A? pathology are detected EMBO Molecular Medicine November 2020
![]() et al., Plasma p-tau181 accurately predicts Alzheimer�s disease pathology at least 8�years prior to post-mortem and improves the clinical characterisation of cognitive decline Acta Neuropathologica July 2020
et al., Plasma p-tau181 accurately predicts Alzheimer�s disease pathology at least 8�years prior to post-mortem and improves the clinical characterisation of cognitive decline Acta Neuropathologica July 2020
![]() et al., Brain Brain March 2023
et al., Brain Brain March 2023
Invasion/Metastasis, Neurobiology, p38 Pathway

 p18INK4C Protein, C32-30G
p18INK4C Protein, C32-30G

 Tau-441 (dK280) Protein, T08-52N
Tau-441 (dK280) Protein, T08-52N

 Tau-441 (dN296) Protein, T08-52BN
Tau-441 (dN296) Protein, T08-52BN

 Tau-441 (T217A) Protein, T08-56ON
Tau-441 (T217A) Protein, T08-56ON
STAY CONNECTED
Fax: 1-604-232-4601